Amy Wagers, Ph.D.

We explore the mechanisms that regulate the function of blood- and muscle-forming and forming stem cells so that their potential can be optimally exploited for the treatment of diseases such as cancer, anemia, muscular dystrophy, and diabetes.

Amy Wagers, Ph.D.
- Forst Family Professor of Stem Cell and Regenerative Biology
- Chair
Harvard Department of Stem Cell and Regenerative Biology - Executive Committee Member
Harvard Stem Cell Institute - Past Co-Chair (2018-2023)
Harvard Department of Stem Cell and Regenerative Biology
Dr. Amy Wagers seeks to change the way we repair our tissues after an injury. Her laboratory’s research focuses on defining the factors and mechanisms that regulate the migration, expansion, and regenerative potential of adult blood-forming and muscle-forming stem cells.
Amy Wagers received her Ph.D. in Immunology and Microbial Pathogenesis from Northwestern University in 1999, and completed her postdoctoral fellowship in the laboratory of Dr. Irving Weissman at Stanford University School of Medicine. In May 2004, she joined the faculty at Harvard Medical School as an Assistant Professor of Pathology and an Investigator at the Joslin Diabetes Center. In 2008, she moved to Harvard’s Department of Stem Cell and Regenerative Biology, and in 2012 became the Forst Family Professor of Stem Cell and Regenerative Biology.
Dr. Wagers is Chair of HSCRB, a Harvard College Professor, and a recipient of a 2018 NIH Pioneer Award for “High-Risk, High-Reward Research”. She has received the Burroughs Wellcome Fund Career Award in the Biomedical Sciences, the Smith Family New Investigator Award, and the Keck Foundation Young Scholars Award.
Wagers’ substantial contributions to science, published in more than 150 primary research and review articles, have brought to light novel regulators (both intrinsic and extrinsic) of stem cell activity in injury repair, degenerative disease, and malignancy, and highlighted key roles for specific blood-borne mediators, cellular niches, inflammatory and metabolic cues in coordinating the functions of stem cells and their progeny throughout the body.
She has also established groundbreaking methods for manipulating stem cell genomes in situ — work that opens new avenues of research and new possibilities for treating congenital and age-related diseases.
In addition to her work at the Harvard Stem Cell Institute, Wagers is a senior investigator in the section on Islet Cell and Regenerative Biology at the Joslin Diabetes Center and a member of the Paul F. Glenn Center for the Biology of Aging at Harvard Medical School.
Lab Overview
Effective functioning of the body’s tissues and organs depends upon innate regenerative processes that maintain proper cell numbers (homeostasis) and replace damaged cells after injury (repair). In many tissues, regenerative potential is determined by the presence and functionality of a dedicated population of stem and progenitor cells, which respond to exogenous cues to produce replacement cells when needed.
Understanding how these unspecialized precursors are maintained and regulated is essential for understanding the fundamental biology of tissues. In addition, this knowledge has practical implications, as the regenerative potential of tissue-specific stem and progenitor cells can be exploited therapeutically by transplantation (to replenish the stem cell pool) or by endogenous manipulation (to boost the repair activity of cells already present in the tissue).
-
Editing stem cells in vivo
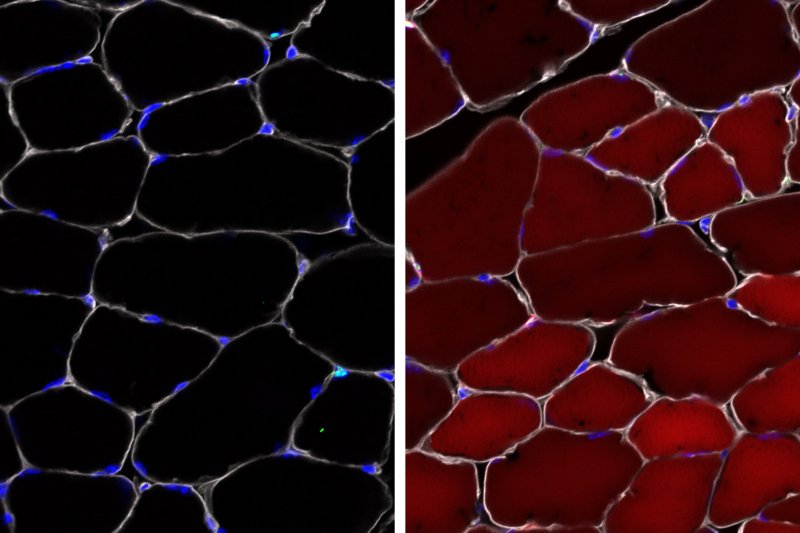
Building on our work in mice with Duchenne muscular dystrophy, our group used an adeno-associated virus (AAV) as a transport vehicle to deliver gene-editing cargo rapidly into several different types of skin, blood, and muscle stem and progenitor cells. We are expanding this work, which has implications for the development of therapies for diseases like spinal muscular atrophy.More about this work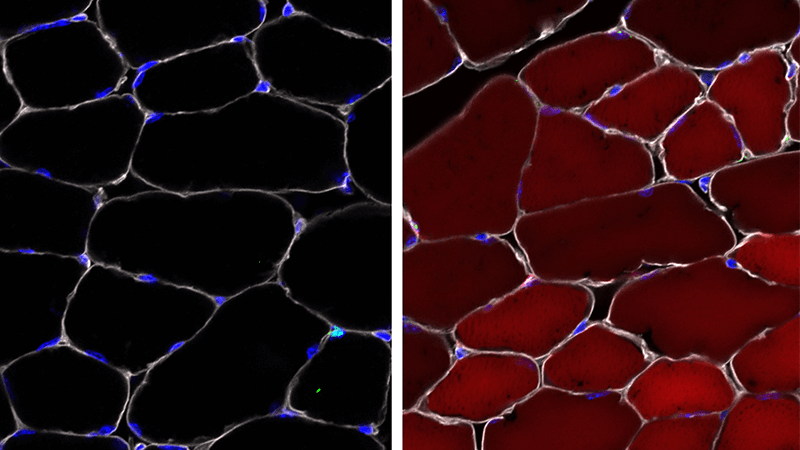
-
Novel regulators of stem cell trafficking
Our studies have identified novel regulators of stem cell trafficking, including the transcription factor EGR1, which uniquely co-ordinates the migration and proliferation of blood stem cells in order to maintain appropriate stem cell number in the bone marrow and to support peripheral immune and inflammatory responses.More about this work -
Marker-based cell sorting to analyze precursor cells
We developed and applied marker-based cell sorting approaches to analyze precursor cells in skeletal muscle, and identified a subset of satellite cells (mononuclear cells found beneath the basal lamina surrounding mature muscle fibers) that act as muscle stem cells. We demonstrated that these unique stem cells exhibit robust self-renewal and myogenic differentiation activity, and can restore muscle function when transplanted into injured or diseased muscle.More about this work