Douglas Melton, Ph.D.

The Melton Lab makes functional islets that secrete insulin to study and cure type-I diabetes. They study the developmental biology of the pancreas, and investigate ways to protect beta cells from autoimmune attack.

Douglas Melton, Ph.D.
- Research Scholar
Vertex Pharmaceuticals - Investigator
Howard Hughes Medical Institute - Co-Director
Harvard Stem Cell Institute - Past Co-Chair (2008-2013)
Harvard Department of Stem Cell and Regenerative Biology
Doug Melton is pursuing a cure for type 1 diabetes. His lab studies the developmental biology of the pancreas, using that information to grow and develop pancreatic cells (islets of Langerhans). In parallel, they investigate ways to protect beta cells from autoimmune attack.
Research in the Melton Lab
The Melton Lab focuses on the developmental biology of the pancreas. We wish to understand how the pancreas normally develops and use that information to grow and develop pancreatic cells (islets of Langerhans). One goal is to understand how vertebrates make an organ from undifferentiated embryonic cells. A longer-term goal has practical significance: if our studies are successful, it should be possible to apply our conclusions to human cells and provide a source of insulin-producing beta-cells for diabetics.
Our main challenge is to understand the precursor or stem cells that give rise to the pancreas and to characterize the key gene products that specify cell fates and functions during organogenesis. To this end, we use several vertebrate organisms, including frogs and chickens but the majority of our studies are done with mice and human embryonic stem cells. We use a wide variety of techniques, including functional genomics, chemical screening, tissue explants and grafting for analyzing inductive signals, and developmental genetics for direct assays of gene function. The aim of all our experiments is to understand the genes, cells, and tissues that direct pancreatic organogenesis.
Biosketch
Melton earned a bachelor’s degree in biology from the University of Illinois and then went to Cambridge University in England as a Marshall Scholar. He earned a BA in history and philosophy of science at Cambridge and remained there to earn a PhD in molecular biology at Trinity College and the MRC Laboratory of Molecular Biology. He is co-director of Harvard’s Stem Cell Institute and an Investigator of the Howard Hughes Medical Institute. He is also a co-founder and scientific advisory board member of Semma Therapeutics, scientific advisory board member of Fidelity Biosciences, and fiduciary board member of Bluebird Bio.
Lab Overview
Our goal is to develop significant new treatments for diabetes. We aim to eliminate the present practice of regular blood checks and insulin injections, replacing them with insulin-producing cell transplants, specifically pancreatic beta cells that measure glucose levels and secrete just the right amount of insulin.
Our approach is best characterized as applying developmental biology to understand and change the course of diabetes.
The methods we have developed to make hundreds of millions of functional beta cells from human stem cells (ES or iPS cells) form the central theme for our research. In one instance, we study how to make all the islet endocrine cells, including alpha (glucagon-producing) and delta (somatostatin-producing) cells and produce islet-like clusters. These human stem cell-derived islet clusters are used both in vitro and in vivo for metabolic studies on islet function.
iPS cells derived from either Type 1 (juvenile) or Type 2 (adult onset) diabetics has made it possible to begin studies on the root cause(s) of diabetes. These stem cell islet clusters, derived using patient’s blood, enable studies on diabetic islet biology, and are being used to understand the cellular and genetic basis of the autoimmune attack in Type 1 diabetes.
-
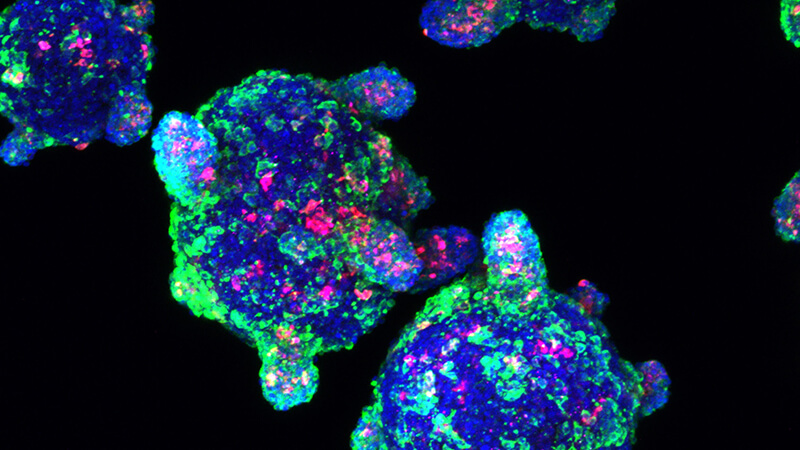
In vitro generation of stem-cell-derived beta cells
We have developed methods to make hundreds of millions of functional beta cells from human stem cells (ES or iPS cells). This allows us to pursue research that will lead to new treatments – or even a cure – for Type 1 diabetes. Currently, we are expanding on this work to study how one could make all the islet endocrine cells, including alpha (glucagon-producing) and delta (somatostatin-producing) cells, and produce islet-like clusters that allow us to carry out metabolic studies on islet function.More about this work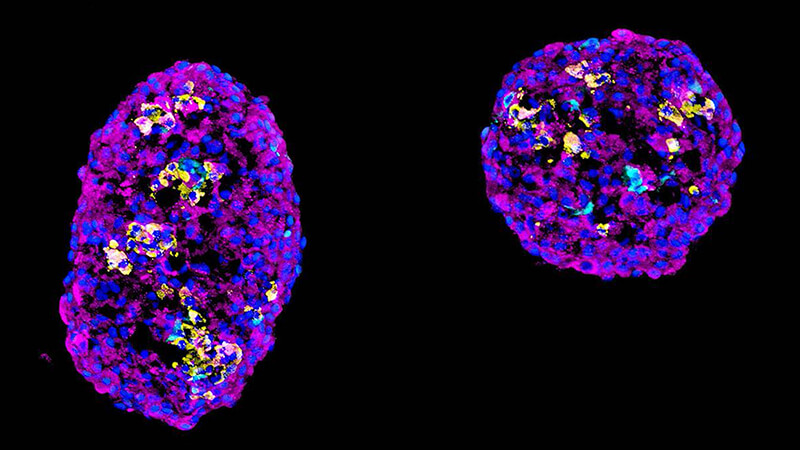
-
Immune modeling of Type I diabetes
We use induced-pluripotent stem (iPS) cells derived from diabetes patients to explore the root cause(s) of the disease. We create clusters of islet cells to study both diabetic islet biology and the cellular and genetic basis of the autoimmune attack in Type 1 diabetes.More about this work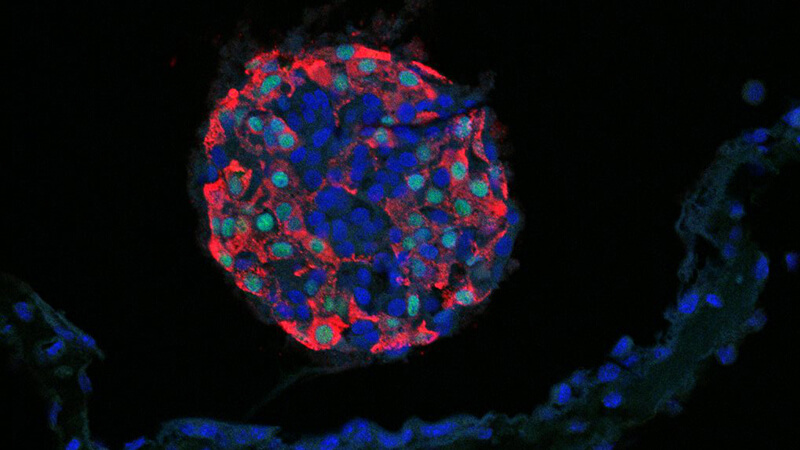
-
Engineering protection of beta cells from the immune system
By genetically modifying stem cells, the starting material for islet clusters, we aim to gain more complete control of in vitro differentiation by altering genes that determine cell fates. In collaboration with bioengineers, we explore physical protection following transplantation. We also genetically alter the islet cells so as to blunt or avoid an immune attack.More about this work