Ya-Chieh Hsu, Ph.D.

Stem cell behaviors are controlled by both local and systemic cues. We aim to discover the principles of cell-cell interactions governing development, regeneration, and injury repair, as well as how systemic changes of the body alter these cell-cell interactions.
We focus on the mammalian skin— an accessible organ with diverse cell types, multiple populations of stem cells, and amenable to both single-cell genomics and rapid in vivo gene editing.

Ya-Chieh Hsu, Ph.D.
- Professor of Stem Cell and Regenerative Biology
- Principal Faculty
Harvard Stem Cell Institute
Ya-Chieh Hsu is Professor of Stem Cell and Regenerative Biology, a Principal Faculty Member at the Harvard Stem Cell Institute, and an associate member of the Broad Institute.
The Hsu laboratory seeks to understand the principles and molecular nature of cell-cell interactions governing development, regeneration, and injury repair using the mammalian skin— an accessible organ with diverse cell types and multiple populations of somatic stem cells.
Dr. Hsu completed her Ph.D. at Baylor College of Medicine, where she studied pathways controlling organ size using Drosophila as a model. For her postdoctoral research, she delineated the lineage hierarchy of hair follicle cells and investigated how signals from stem cell progeny regulate hair follicle stem cells in Elaine Fuchs’ laboratory at the Rockefeller University.
Dr. Hsu is a recipient of several honors and awards, including the Pew Biomedical Scholars Award, the Smith Family Award for Excellence in Biomedical Research, Basil O’Connor Starter Scholar Award, Smith Family Foundation Odyssey Award, and American Cancer Society Research Scholar Award. She is also an awardee of Harvard’s Roslyn Abramson Award for excellence and sensitivity in undergraduate teaching.
Lab Overview
Mammalian skin serves as a physical barrier protecting organisms from injury, infection, and dehydration. The skin also regulates body temperature and receives complex sensory inputs. These diverse functions are made possible by a rich array of cell types. The epidermis, the hair follicle, and the melanocyte lineage contain tissue-resident stem cells and are among some of the most highly regenerative tissues in adult mammals. These stem cells regenerate in a rich environment filled with fibroblasts, immune cells, neurons, blood vessels, muscle, and adipocytes. The cell-cell interactions that occur in this organ are complex and vast in number. Therefore, the mammalian skin provides us with a plethora of opportunities to explore how cells from diverse lineages coordinate their behaviors to build and maintain a functional organ.
-
The Cellular and Molecular Identity of the Stem Cell Niche
Stem cell behaviors are heavily influenced by the niche microenvironment, where stem cells reside. However, the identity of niche factors and niche cell types remains elusive for many mammalian stem cells. We have established tools and strategies to manipulate gene expression in numerous skin cell types and are using them to investigate reciprocal interactions between stem cells and niches. This research will define mechanisms that could be targeted to promote tissue regeneration or wound healing.More about this work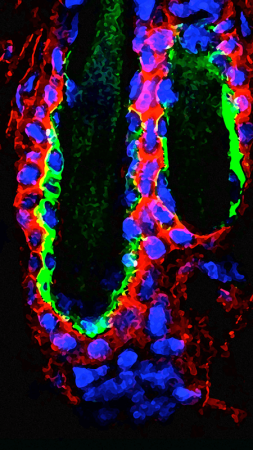
-
Function and Biology of Transit-Amplifying Cells
Somatic stem cells are often quiescent but occasionally divide to generate “transit-amplifying cells” (TACs). TACs then undergo rapid division and expansion to produce a large quantity of differentiated cells downstream. Since TACs are the workforce of tissue production while stem cells are mostly dormant, TACs are located at an ideal juncture to orchestrate production of their own downstream progeny with critical changes in surrounding niche cell types that accommodate and support a regenerating tissue (see our review comparing TACs in the blood and the skin (Zhang and Hsu WIREs Developmental Biology).More about this work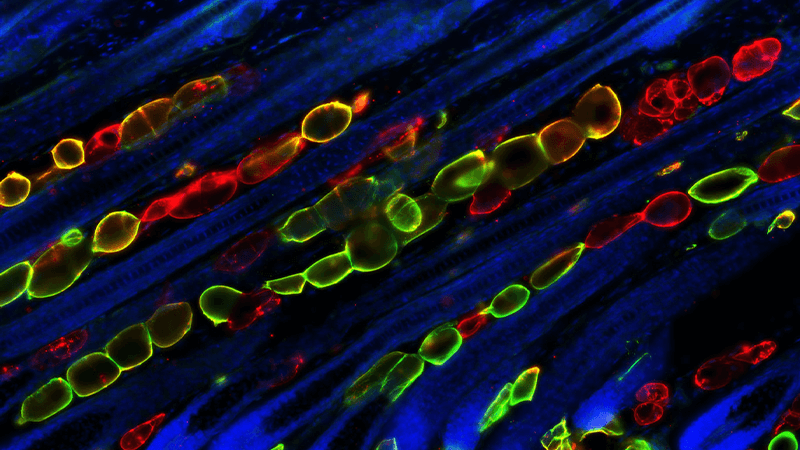
-
How Systemic Changes Regulate Stem Cell Behavior
Stem cells must respond not just to the niche but to systemic signals in response to stress, pregnancy, or aging. We have established approaches to determine if and how changes in systemic factors influence cell-cell interactions, stem cell behavior, and repair of injury in the skin. We have defined several circulating factors crucial for skin stem cell function and are determining whether these factors act directly or through the niche to regulate stem cell activity and tissue regeneration.