
Blood stem cells, which give rise to all of our blood cell types, undergo a quality assurance process after they’re born. As the lab of Leonard Zon, MD, Professor of Stem Cell and Regenerative Biology and director of the Stem Cell Research program at Boston Children’s, has documented, immune cells known as macrophages interact with each newly born cell. They then engulf and eat those that are mutated or diseased, cued by a surface molecule called calreticulin, while encouraging healthy stem cells to divide and proliferate.
Now, studying zebrafish, researchers in the Zon Lab show in the journal Science that calreticulin — the “eat me” signal — can be induced on blood stem cells in several ways, as can a complementary “don’t eat me” signal the lab identified, beta-2-microglobulin (B2m).
They also found ways to manipulate the two signals, potentially preventing mutant, cancerous stem-cell populations or “clones” from taking over.
“We want to educate macrophages to take out the mutant stem cells,” says Zon, who is also affiliated with the Dana-Farber/Boston Children’s Cancer and Blood Disorders Center. “They do take out the mutants periodically, but they’re not efficient and a cancer can overwhelm the system. This could be a whole new way of treating blood cancers like leukemia.”
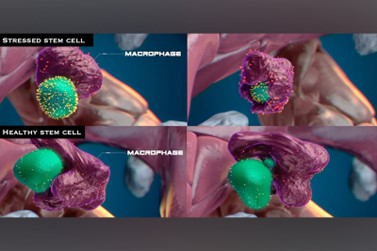
Finding the signals of blood stem cells
To better understand the eat-me signal’s origins, Zon and colleagues screened a panel of 1,200 chemicals in human cells and found 93 compounds that induced production of calreticulin on their surface.
Just over half of the compounds induced calreticulin by increasing cellular levels of reactive oxygen species (ROS), known indicators of cellular stress. But the remaining compounds induced calreticulin through pathways that did not involve ROS. Live zebrafish assays, using live imaging and cellular barcoding techniques, revealed that macrophages killed the stem cells with increased ROS, allowing the rest to proliferate.
Separately, the team conducted a whole-genome CRISPR screen in human leukemic cells, in which they knocked out more than 10,000 genes to identify those that influenced whether macrophages killed blood stem cells or let them live. Together with the chemical screen and the genomic approach, the findings suggested there might be an active don’t-eat-me signal.
The experiments pointed to the gene encoding TLR3, a receptor in the innate immune system. When it was knocked out, macrophages killed more blood stem cells. When TLR3 signaling was left intact, B2m — the don’t-eat-me signal — went to the cell surface.
“When we made fish without B2m, more blood stem cells were engulfed and eliminated,” says Cecilia Pessoa Rodrigues, PhD, of the Zon lab and Harvard Stem Cell Institute, first author on the Science paper.
The team found another key player: RNA repetitive elements. These elements are scattered around the genome, and the body normally tries to suppress them. Zon’s team showed that de-repressing these elements with methylation inhibitors triggered the TLR3 gene and increased B2m expression on the cell surface, enabling more blood stem cells to survive.
Tipping blood stem cells away from cancer
This work points to several prospects for controlling blood cancers by tweaking blood stem cells’ balance of calreticulin (the eat-me signal) and B2m (the don’t-eat-me signal). Drugs boosting calreticulin on the cell surface could possibly be combined with an antibody blocking B2m or TLR3. Or either approach might work on its own.
The Zon lab is now screening for drugs that create an eat-me signal in mutant stem cells but not in normal stem cells. They’re also looking for drugs that will remove mutant clones of cells as a new treatment for preleukemia and leukemia.
In addition to treating cancer, Zon envisions using these approaches to treat clonal hematopoiesis. In this precancerous condition, which becomes more common as people age, acquired mutations can cause mutant blood stem cell populations to multiply faster than others, eventually taking up a large share of a person’s blood cells.
“I think you can manipulate the system in multiple ways to get rid of bad stem cell clones and keep good clones,” Zon says. “Getting rid of mutant clones could prevent cancer down the road.”